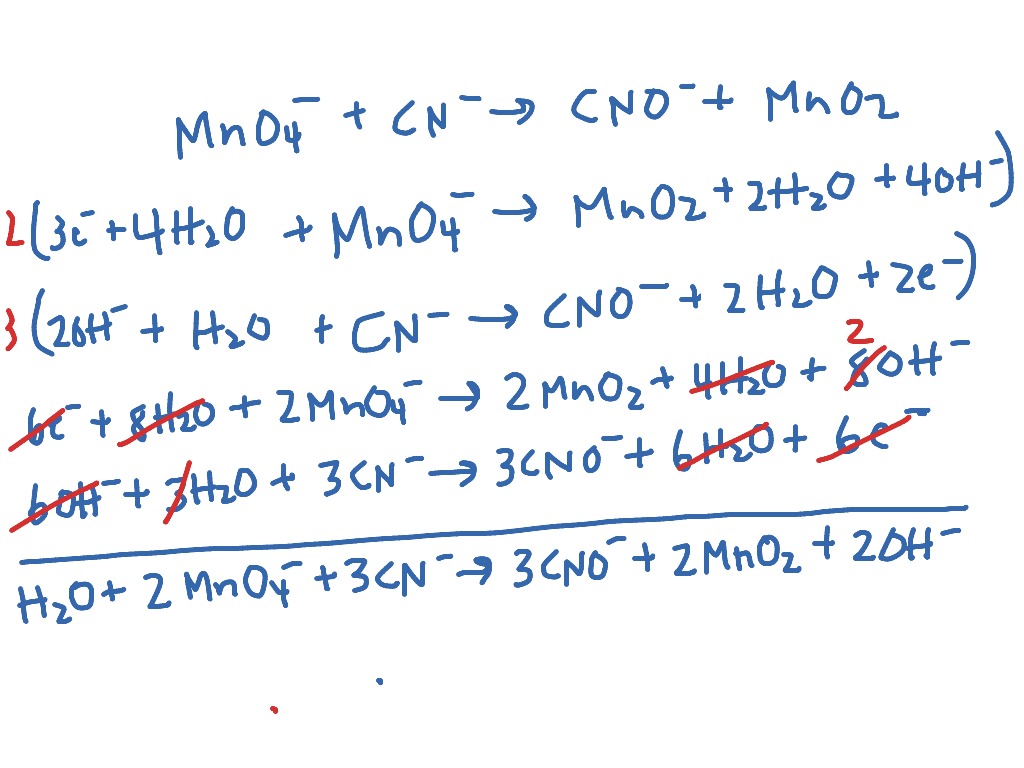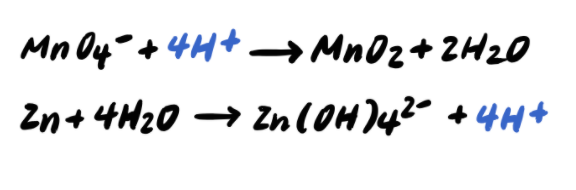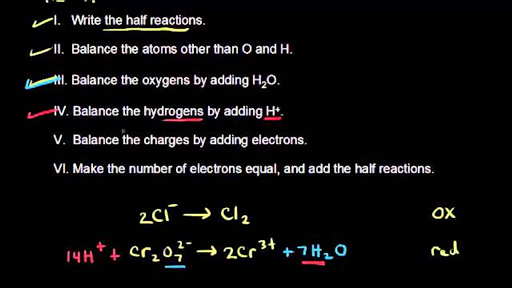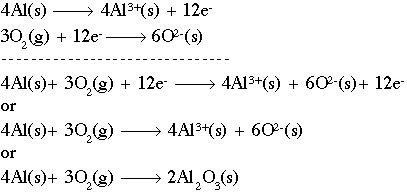Balancing Redox Reactions: Acid Conditions Balance the following redox equation: Cr 2 O 7 2- (aq) + HNO 2 (aq) --> Cr 3+ (aq) + NO 3 - (aq) (acidic) Oxidation. - ppt download

Balancing redox equations | Science, Chemistry, Balancing Equations, Balancing Redox, Redox Reactions | ShowMe