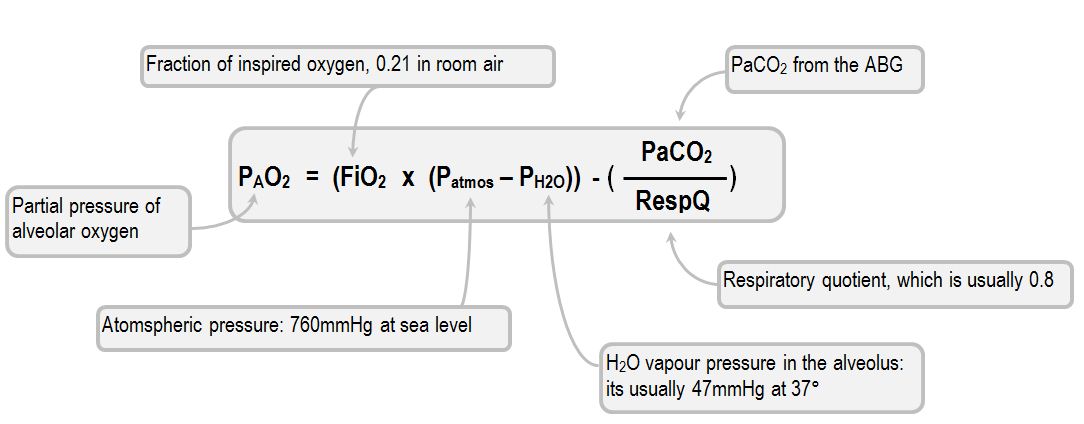
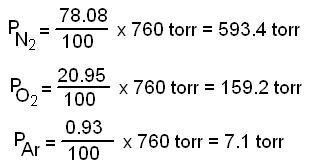Relating oxygen partial pressure, saturation and content: the haemoglobin– oxygen dissociation curve | European Respiratory Society

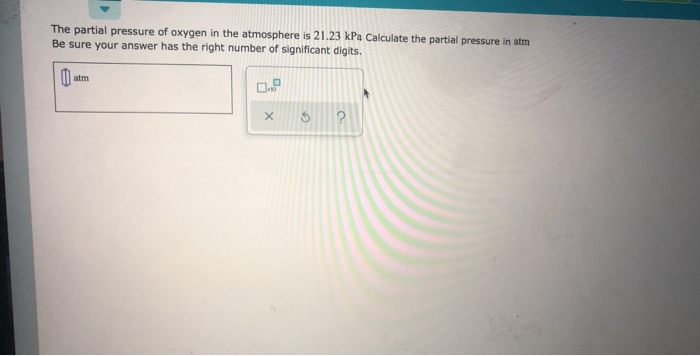
SOLVED: Calculate the partial pressures (in atmospheres) of both nitrogen and oxygen in Mike's blood at a depth of 220 feet and at sea level. Assume the SCUBA tank contains air. Information

A temperature-partial pressure of oxygen diagram calculated by FactSage... | Download Scientific Diagram

SOLVED: Calculate the partial pressure of each of the following gases in room. Air when the barometric pressure is 760 mmHg ( assume the room air contains 21% oxygen, 78% nitrogen, and