
Calculate the molarity `(M)` and normality `(N)` of a solution of oxalic acid `[(COOH)_(2) . - YouTube

Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube

SOLVED: Calculate the percent mass per volume, % (m/v), of a dextrose solution containing 6.50 g of dextrose in 2.50×102 mL of solution. Note that mass is not technically the same as

📐Calculate the slope of the line on the graph using the formula: m = m = v subscript 2 - v subscript - Brainly.com

SOLVED: 3. Calculate the molarity ofacetic acid using the volume of vinegar solution used in the titration This is the molarity of 0.5% (w/v) vinegar solution: moles M= 1 83*lo meles M -
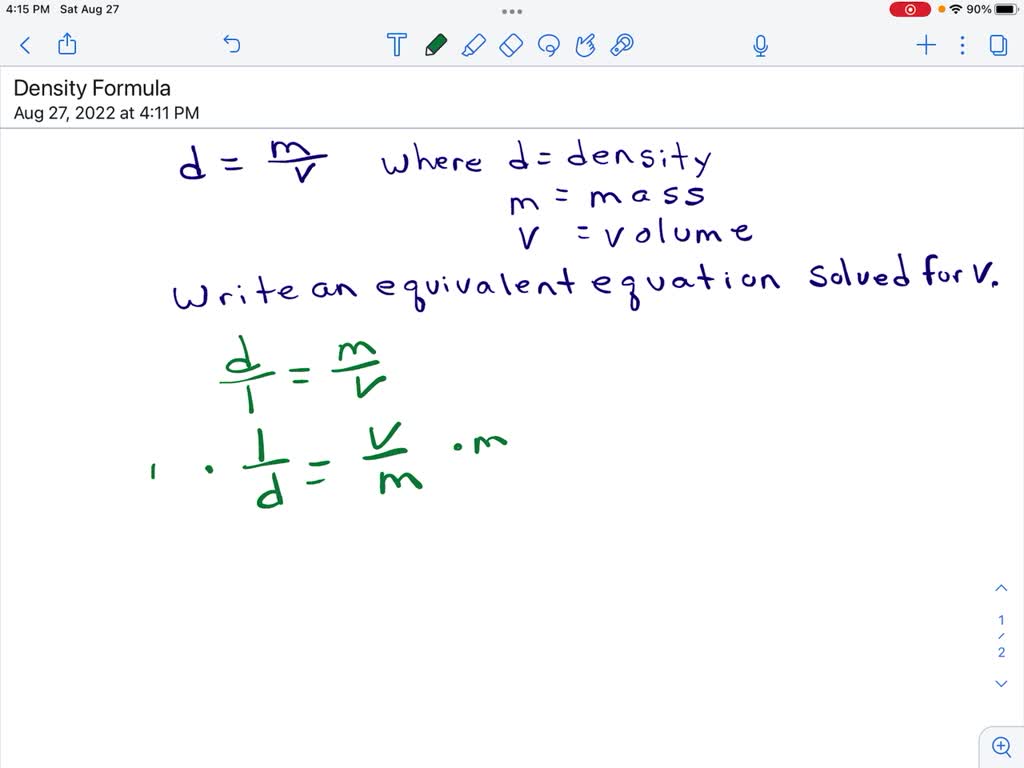
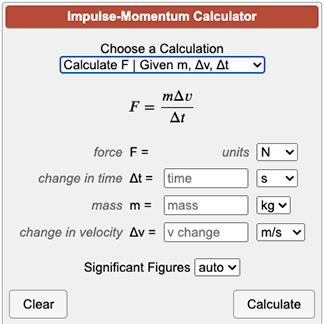
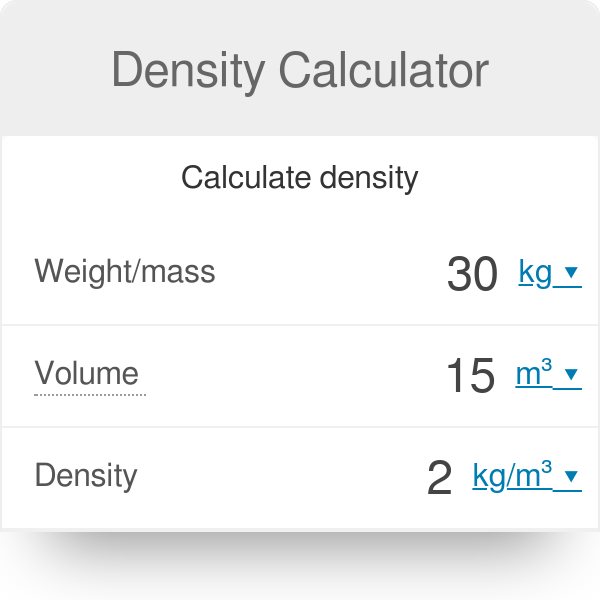


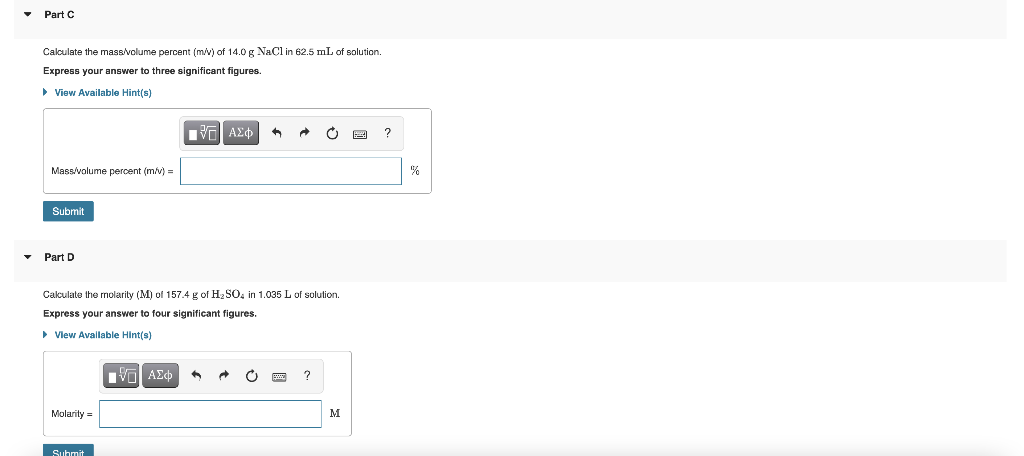


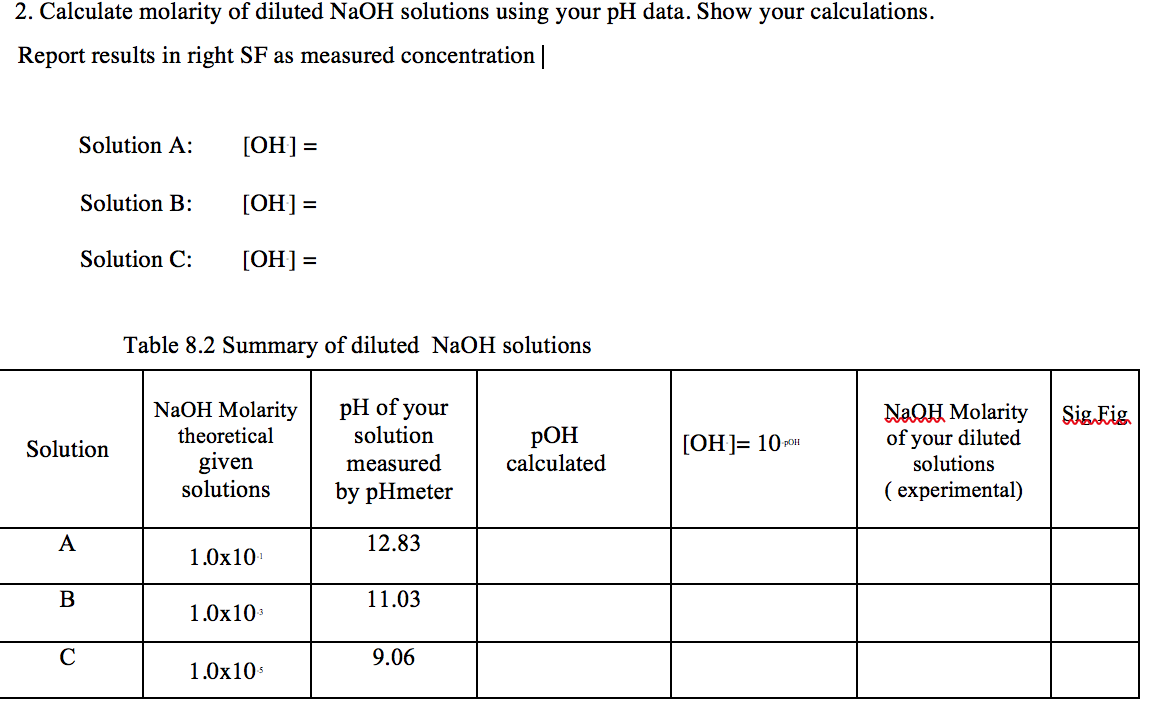



![Molarity Calculator [with Molar Formula] Molarity Calculator [with Molar Formula]](https://scrn-cdn.omnicalculator.com/chemistry/molarity@2.png)



