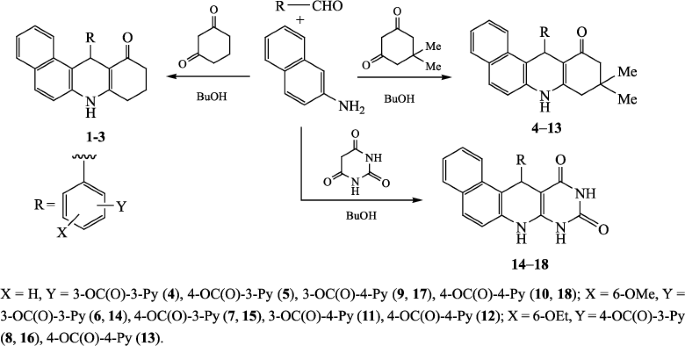
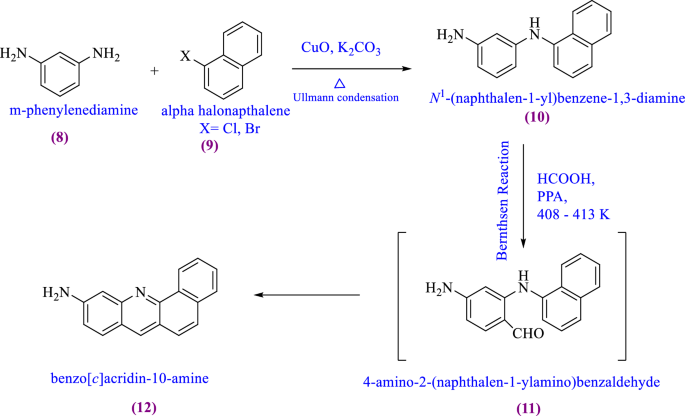
US10937976B2 - Tetradentate platinum and palladium complex emitters containing phenyl-pyrazole and its analogues - Google Patents

Synthesis and Spectrophotometric Studies of 9‐Substituted‐4,5‐dimethoxyacridine Multifunctionalizable Fluorescent Dyes and Their Macrocyclic Derivatives - Golcs - 2021 - European Journal of Organic Chemistry - Wiley Online Library

A new synthesis strategy for acridine derivatives to constructing novel host for phosphorescent organic light-emitting diodes - ScienceDirect
![Molecules | Free Full-Text | Grindstone Chemistry: Design, One-Pot Synthesis, and Promising Anticancer Activity of Spiro[acridine-9,2′-indoline]-1,3,8-trione Derivatives against the MCF-7 Cancer Cell Line Molecules | Free Full-Text | Grindstone Chemistry: Design, One-Pot Synthesis, and Promising Anticancer Activity of Spiro[acridine-9,2′-indoline]-1,3,8-trione Derivatives against the MCF-7 Cancer Cell Line](https://www.mdpi.com/molecules/molecules-25-05862/article_deploy/html/images/molecules-25-05862-g001.png)
Molecules | Free Full-Text | Grindstone Chemistry: Design, One-Pot Synthesis, and Promising Anticancer Activity of Spiro[acridine-9,2′-indoline]-1,3,8-trione Derivatives against the MCF-7 Cancer Cell Line

US20220169607A1 - Carbazole and acridine photoredox catalysts for small molecule and macromolecular transformations - Google Patents

PDF) Synthesis, Spectroscopic, Crystal Structures, and Potentiometric Studies of a Proton Transfer Compound of Acridine with Pyridine-2,6-dicarboxylic Acid and a Novel Palladium(II) Complex with Coordination of Acridine

Shining Visible Light on Reductive Elimination: Acridine–Pd-Catalyzed Cross-Coupling of Aryl Halides with Carboxylic Acids | Journal of the American Chemical Society

PDF) The Synthesis of a Novel Anticancer Compound, N-(3,5 Dimethoxyphenyl) Acridin-9-Amine and Evaluation of Its Toxicity
![Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2666086523000024-sc2.jpg)
Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect

Fluorescent images of acridine orange/ ethidium bromide dual stained... | Download Scientific Diagram
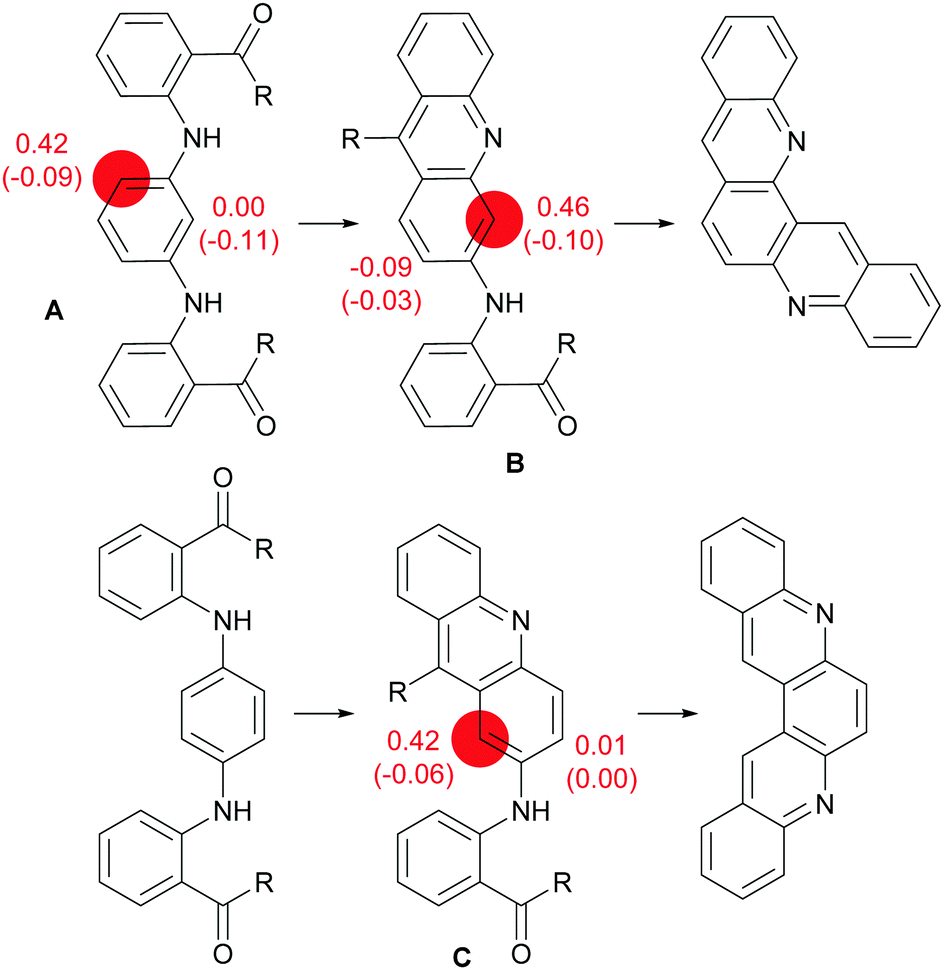
Aza-aromatic polycycles based on triphenylene and acridine or acridone: synthesis and properties - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ02630E
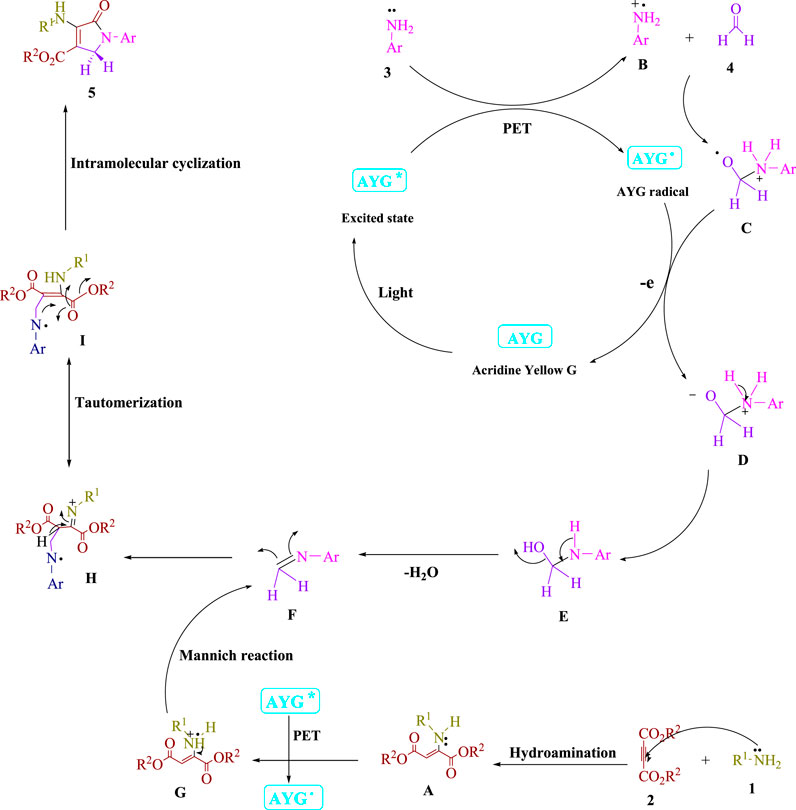
Frontiers | Acridine yellow G (AYG) as a photo-induced electron transfer (PET) photocatalyst employed for the radical Michael–Mannich cyclocondensation of imines
![Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2666086523000024-gr1.jpg)
Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect
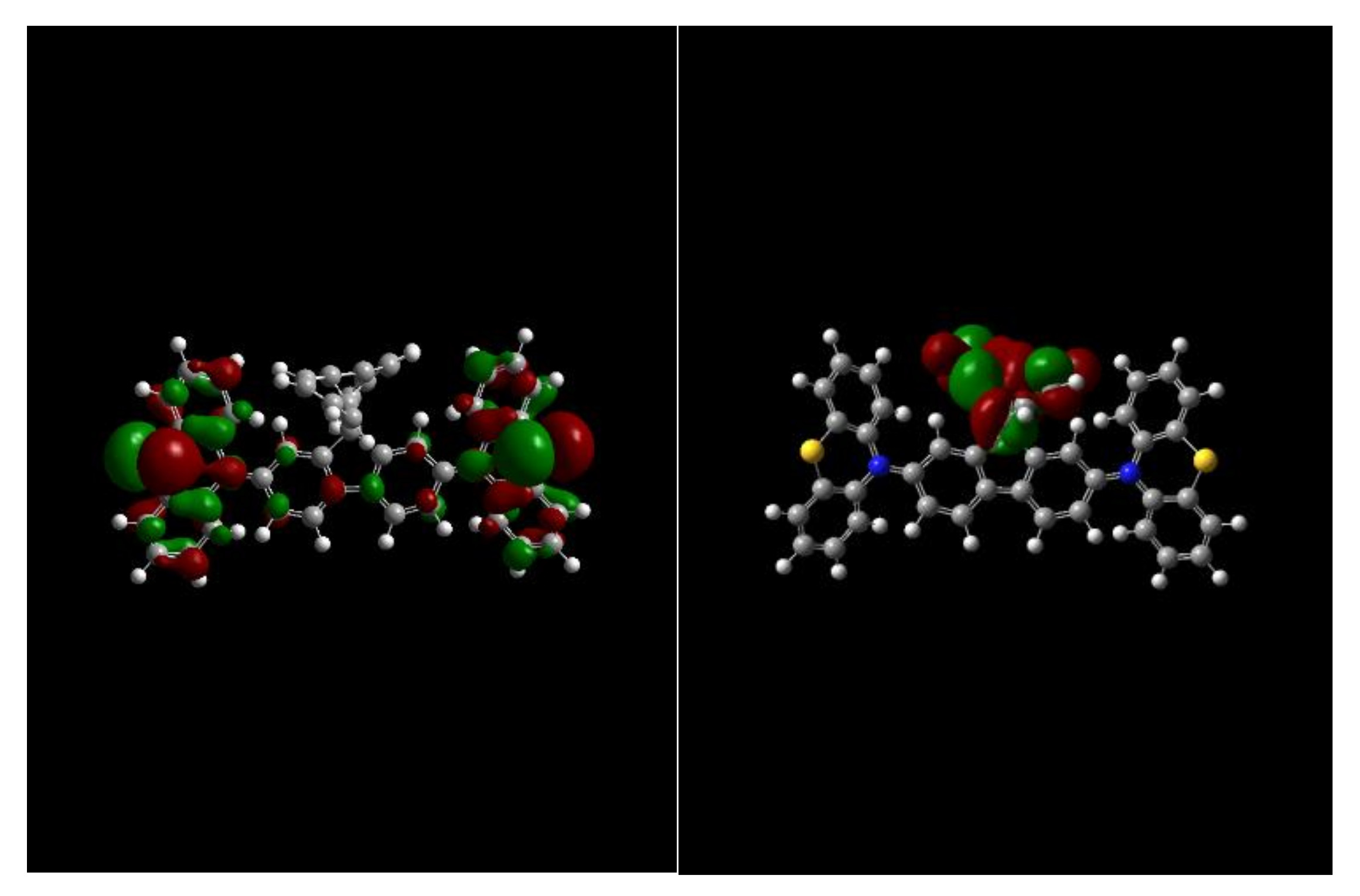
Molecules | Free Full-Text | Utilizing a Spiro Core with Acridine- and Phenothiazine-Based New Hole Transporting Materials for Highly Efficient Green Phosphorescent Organic Light-Emitting Diodes
![6,5] CNN Palladium(II) Pincer Complexes Containing N-Substituted Monoanionic and Dianionic Guanidinate Ligands: Syntheses, Structural Aspects, and Their Utility in Suzuki–Miyaura Coupling Reactions | Organometallics 6,5] CNN Palladium(II) Pincer Complexes Containing N-Substituted Monoanionic and Dianionic Guanidinate Ligands: Syntheses, Structural Aspects, and Their Utility in Suzuki–Miyaura Coupling Reactions | Organometallics](https://pubs.acs.org/cms/10.1021/acs.organomet.1c00414/asset/images/large/om1c00414_0026.jpeg)
6,5] CNN Palladium(II) Pincer Complexes Containing N-Substituted Monoanionic and Dianionic Guanidinate Ligands: Syntheses, Structural Aspects, and Their Utility in Suzuki–Miyaura Coupling Reactions | Organometallics
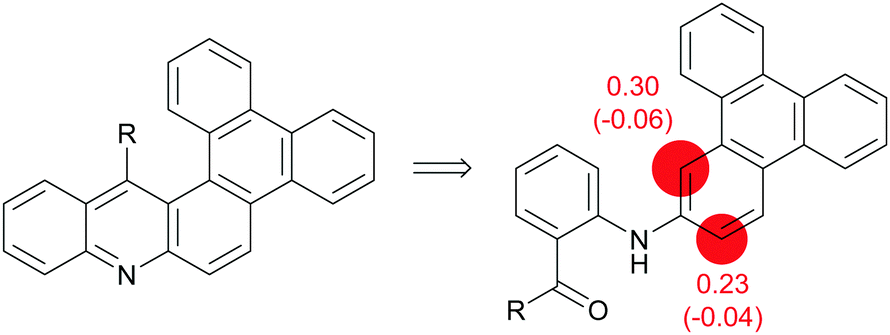
Aza-aromatic polycycles based on triphenylene and acridine or acridone: synthesis and properties - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ02630E
![Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S2666086523000024-fx3b.jpg)
Acridine yellow G as a photo-induced electron transfer catalyzed radical metal-free synthesis of tetrahydrobenzo[b]pyran scaffolds in an aqueous media - ScienceDirect

![92638-81-8|10H-Spiro[acridine-9,9'-fluorene]| Ambeed 92638-81-8|10H-Spiro[acridine-9,9'-fluorene]| Ambeed](https://file.ambeed.com/static/upload/proimg/333/92638-81-8.png)